MEET NEX
NEXVIAZYME fast facts
NEXVIAZYME is an enzyme replacement monotherapy† for people new to treatment or those switching therapies.
†Not including premedication or pretreatment your doctor may prescribe.
Switching to NEXVIAZYME can be a seamless process with the help of CareConnectPSS®.
FIND OUT
How does NEXVIAZYME work?
NEXVIAZYME is an enzyme replacement therapy (ERT) that helps provide more of the enzyme people with LOPD need to help treat this degenerative muscle disease.
People with LOPD do not have enough GAA enzyme. Without GAA, glycogen builds up, causing ongoing muscle damage.
In a normal muscle cell, the GAA enzyme breaks down glycogen, a type of sugar, so the cell can use it for energy when needed. To get to the muscle cell, GAA needs the help of M6P, a part of a molecule.
NEXVIAZYME helps supply more of the GAA enzyme that muscle cells need to function properly.
NEXVIAZYME is an M6P-enriched ERT for LOPD and is scientifically designed to be absorbed effectively by muscle cells.
What is M6P?
M6P (mannose-6-phosphate) is part of the molecule that binds to certain receptors on the muscle cell and helps enzyme replacement therapy reach your muscle cells.
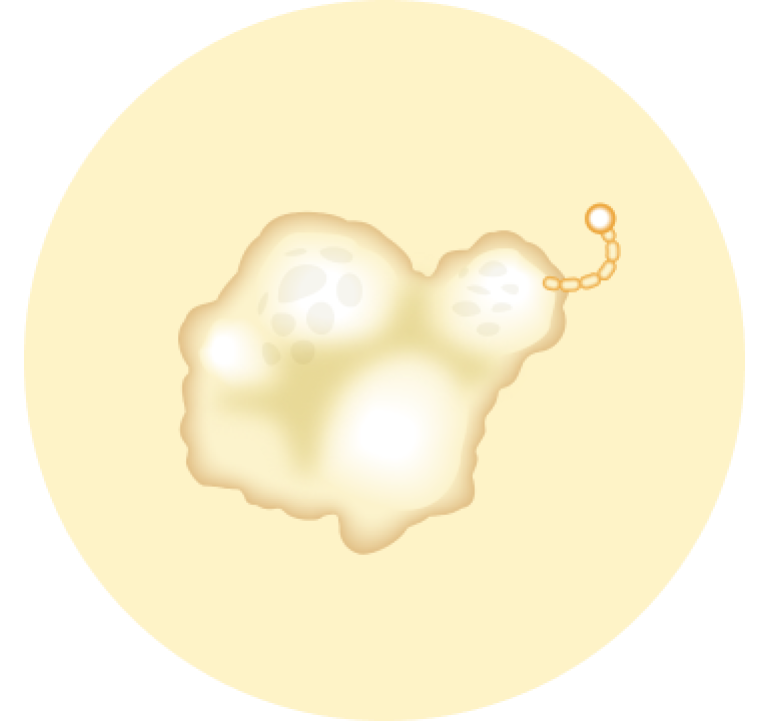
NEXVIAZYME has ~15x more M6P than alglucosidase alfa (Lumizyme) and was made to improve uptake of the treatment into the muscle cells.
NEXVIAZYME
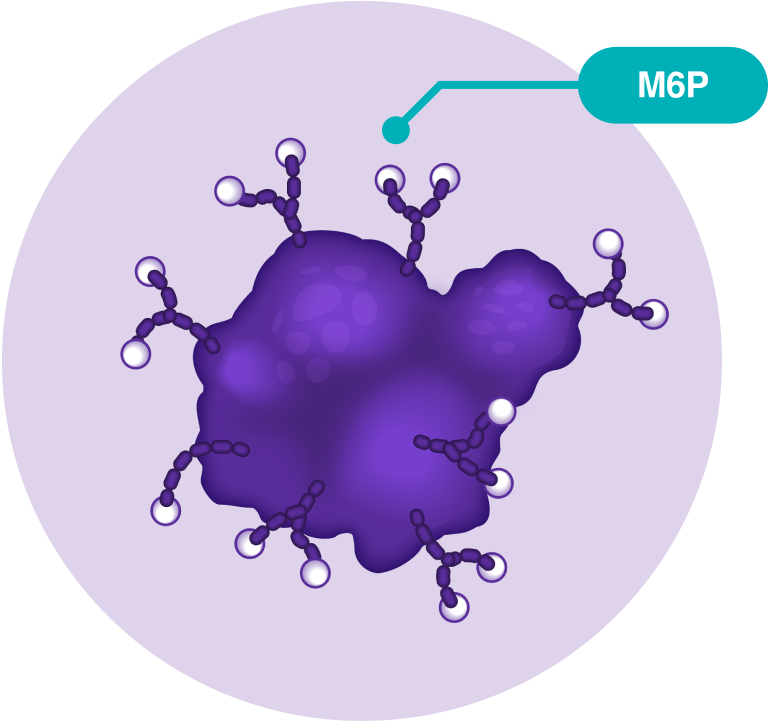
alglucosidase alfa
Join what’s NEX
Sign up for Pompe news, community support, and more.
SEE THE PROOF
Look at the improvement patients saw
NEXVIAZYME vs alglucosidase alfa
NEXVIAZYME showed meaningful improvement for breathing and walking when compared to alglucosidase alfa in the COMET trial.
This is how the study was set up:
- Everyone (100 patients ages 16-78) in the study had LOPD, but none had prior treatment.
- Some were given NEXVIAZYME, others were given alglucosidase alfa.
- Everyone stayed in the study for 49 weeks.
- Two tests were used to measure how people were doing at the beginning and at the end of the study:
FORCED VITAL CAPACITY (FVC)
measures how much air you can force out in a deep breath
6-MINUTE WALK TEST (6MWT)
measures the distance you can walk in 6 minutes
After ~1 year (49 weeks) on NEXVIAZYME, people were able to:
Breathe Easier
by an average of 2.9 percentage points in a breathing test*
*Compared to when they began treatment.
- People taking alglucosidase alfa saw improvements in breathing by an average of 0.5 percentage points.
- People treated with NEXVIAZYME saw an improvement of 2.4 percentage points compared with people treated with alglucosidase alfa, although NEXVIAZYME was not statistically superior.

Walk Farther
by an average of 106 feet during a 6MWT*
*Compared to when they began treatment
- People taking alglucosidase alfa improved their walking distance by an average of 7.2 feet.
- People taking NEXVIAZYME walked an average of 98 feet farther than those taking alglucosidase alfa. This measurement was not tested to determine statistical superiority of NEXVIAZYME to alglucosidase alfa.
What happened NEX? Long-term stability
Following the COMET study (end of week 49), two groups of patients were followed for approximately two additional years (145 weeks total). We found that people who started NEXVIAZYME, as well as people who switched to NEXVIAZYME at week 49, all maintained stability in breathing and walking compared to week 0.
NEXVIAZYME patients maintained stability in breathing and walking


*Prescription data as of March 2025.
†Not including premedication or pretreatment.
The open-label extension study:*
After the ~1 year (49 weeks) of the study, 44 people on alglucosidase alfa switched to NEXVIAZYME and the 51 people on NEXVIAZYME continued to take it. After ~3 years (145 weeks) of the study, people who started or switched to NEXVIAZYME maintained their breathing and walking abilities.
*Open-label: Both doctors and patients knew what treatment people were taking.
OVER THE YEARS
What was learned about safety
NEXVIAZYME has a demonstrated safety profile with both newly treated patients and those
who switched from alglucosidase alfa.
.png 215w)
Results
In the COMET trial, NEXVIAZYME demonstrated similar safety and risks to alglucosidase alfa. Over the 49-week study, people taking NEXVIAZYME had fewer serious side effects than those taking alglucosidase alfa;† no one had to stop taking NEXVIAZYME because of side effects, while 4 people had to stop taking alglucosidase alfa.
.png 214w)
Antibodies
When antibodies form, they can interfere with some medicines. There is a potential to develop antibodies, but for patients in the trial it had no apparent effect on their efficacy. Patients in the clinical trials who did have high titers for antibodies had a higher risk of experiencing infusion-associated and hypersensitivity reactions.
NEXVIAZYME has nearly 7 years of safety data including people who switched from alglucosidase alfa.
†The COMET trial was not designed to demonstrate a statistically significant difference in the incidence of adverse reactions between NEXVIAZYME and alglucosidase alfa. In addition, 13 out of 51 people (25%) receiving NEXVIAZYME experienced mild to moderate infusion-associated reactions. These reactions included headache, diarrhea, itching, hives, and rash. No one experienced a severe infusion-associated reaction. 1 (2%) patient receiving NEXVIAZYME reported a serious adverse reaction; 3 (6%) patients receiving alglucosidase alfa reported serious adverse reactions.
Common side effects of NEXVIAZYME include:
• headache
• fatigue
• dizziness
• nausea
• diarrhea
• vomiting
• joint pain
• muscle pain
• itching
• shortness of breath
• rash
• “pins-and-needles” sensation
• hives
These are the most common side effects, but others are possible. Always tell your doctor about any changes in the way you feel. You can also report side effects at 1-800-FDA-1088 or www.fda.gov/medwatch.
|
WARNING: SEVERE HYPERSENSITIVITY REACTIONS, INFUSION-ASSOCIATED REACTIONS, and RISK OF ACUTE CARDIORESPIRATORY FAILURE IN SUSCEPTIBLE PATIENTS |
WARNINGS AND PRECAUTIONS
Hypersensitivity Reactions Including Anaphylaxis: See Boxed WARNING. Your doctor may decide to give you antihistamine, anti-fever and/or steroid medications before your infusions. Your doctor should consider the risks and benefits of restarting the infusion if you have a severe hypersensitivity reaction (including anaphylaxis) to NEXVIAZYME. If a mild or moderate hypersensitivity reaction occurs, your healthcare provider may slow the infusion rate or temporarily stop the infusion.
Infusion-Associated Reactions (IARs): See Boxed WARNING. Your doctor may decide to give you medications before your infusions to decrease the risk of IARs; however, IARs may still occur after receiving these medications. If mild or moderate IARs occur, your healthcare provider should consider decreasing the infusion rate or temporarily stopping the infusion which may help improve the symptoms.
Risk of Acute Cardiorespiratory Failure in Susceptible Patients: See Boxed WARNING.
ADVERSE REACTIONS
The most common adverse reactions (>5%) were headache, fatigue, diarrhea, nausea, joint pain, dizziness, muscle pain, itching, vomiting, shortness of breath, rash, “pins-and-needles” sensation, and hives.
INDICATION
NEXVIAZYME (avalglucosidase alfa-ngpt) is used for the treatment of patients 1 year of age and older with late-onset Pompe disease [lysosomal acid alpha-glucosidase (GAA) deficiency].
Please see full Prescribing Information for complete details, including Boxed WARNING.
|
WARNING: SEVERE HYPERSENSITIVITY REACTIONS, INFUSION-ASSOCIATED REACTIONS, and RISK OF ACUTE CARDIORESPIRATORY FAILURE IN SUSCEPTIBLE PATIENTS |
WARNINGS AND PRECAUTIONS
Hypersensitivity Reactions Including Anaphylaxis: See Boxed WARNING. Your doctor may decide to give you antihistamine, anti-fever and/or steroid medications before your infusions. Your doctor should consider the risks and benefits of restarting the infusion if you have a severe hypersensitivity reaction (including anaphylaxis) to NEXVIAZYME. If a mild or moderate hypersensitivity reaction occurs, your healthcare provider may slow the infusion rate or temporarily stop the infusion.
Infusion-Associated Reactions (IARs): See Boxed WARNING. Your doctor may decide to give you medications before your infusions to decrease the risk of IARs; however, IARs may still occur after receiving these medications. If mild or moderate IARs occur, your healthcare provider should consider decreasing the infusion rate or temporarily stopping the infusion which may help improve the symptoms.
Risk of Acute Cardiorespiratory Failure in Susceptible Patients: See Boxed WARNING.
ADVERSE REACTIONS
The most common adverse reactions (>5%) were headache, fatigue, diarrhea, nausea, joint pain, dizziness, muscle pain, itching, vomiting, shortness of breath, rash, “pins-and-needles” sensation, and hives.
INDICATION
NEXVIAZYME (avalglucosidase alfa-ngpt) is used for the treatment of patients 1 year of age and older with late-onset Pompe disease [lysosomal acid alpha-glucosidase (GAA) deficiency].
Please see full Prescribing Information for complete details, including Boxed WARNING.

